Interestingly, the most important part of this announcement is likely the related personnel side of these acquisitions as two world-class experts in hand-portable Polymerase Chain Reaction (PCR) science are joining the Co-Diagnostics team
Two weeks ago, while Yours Truly was off in the middle-of-nowhere Utah spending valuable away time with the 22 most important people in his life (aka, my family), apparently some Utah-based businesses were still working – Christmas season notwithstanding. And getting important things accomplished.
One such company was Salt Lake City-based Co-Diagnostics (NasdaqCM:CODX) which distributed a news release on December 22 announcing that it was acquiring two product development partners for a combined total of nearly 4.72 million shares of Co-Diagnostics common stock (aka, Co-Dx).

The two companies being acquired are
- Advanced Conceptions, and
- Idaho Molecular,
both of which apparently share the same offices in South Salt Lake, Utah. {NOTE: This is actually not surprising given that both have been working in concert with each other and Co-Dx since mid-February 2021.}
According to Co-Dx filings with the U.S. Securities and Exchange Commission for both the Advanced Conceptions acquisition and the Idaho Molecular acquisition, their joint efforts have been centered around
"... the design, engineering and development of a prototype highly sensitive and rapid response home saliva and/or nasal swab diagnostic platform and receptacle kits for, among other things, polymerase chain reaction (PCR) testing and pathogen detection for the ultimate manufacturing, commercialization and marketing of home use, consumer diagnostic kits."
Phew!
According to the SEC filings, each company is being acquired for equal share amounts of common stock of Co-Diagnostics, with
- 1.65 million shares paid outright to the owners of each firm at the concurrent closing of the mergers, and
- 708,500 shares payable to the owners of both of the two firms as certain milestones are achieved in the future, for
- A combined total of nearly 4.72 million shares of CODX.
According to the news release, and confirmed yesterday by Andrew Benson, the Co-Dx Head of Investor Relations, the acquisitions are on track to be consummated before the end of 2021. Yup, before the end of the day this Friday.
Based upon a price of $9.30/share at market close yesterday, that would mean that Co-Dx was paying just under $22 million for each firm (presuming the respective milestones are hit and not including the value of the warrants the owners of both firms could receive and exercise, set at 232,500 warrants each).
{NOTE: "Strike price" figures for the exercise of these warrants was omitted from the respective SEC filings as Co-Dx considers such information as "not material" and "private and confidential."}
According to its acquisition filings with the SEC, Co-Dx currently has 29.16 million shares issued and outstanding, giving the company a valuation this morning before market opening of just over $270 million.
By my analysis, that market capitalization takes into account the two acquisitions as the stock closed at $9.04/share the day before the acquisitions were announced.
The Development Work of Advanced Conceptions and Idaho Molecular Summarized
Since its formation in 2013, Co-Diagnostics has focused on disrupting the molecular diagnostics marketplace by developing smaller, less expensive, and easier to use testing, analysis, and detection systems that deliver fast results (e.g., 30 minutes or less).
As it did for other companies developing and selling similar diagnostics systems and products, the Covid-19 pandemic has clearly benefited Co-Dx.
In fact, as of today, Co-Dx has sold its various tests and products in over 50 countries around the world, many of which have focused on Covid-19.
For in its most recent quarter (ended September 30), Co-Diagnostics generated revenue of over $30 million, a nearly 50% increase from the 3rd quarter in 2020.
On its current trajectory, Co-Dx is likely to surpass $100 million in annual revenue for 2021.
But here's where it gets interesting, because Co-Dx management recognizes that its long-term opportunities lie not with Covid-19, but instead with the plethora of other diseases that torment humans.
Case in point, in a September 21, 2021 video presentation found on the company website, Co-Diagnostics' CEO, Dwight Egan, shares information about its initial success with its still-in-development YouTest PCR system. {NOTE: You can also find this particular video on Vimeo here: https://vimeo.com/626826402?embedded=true&source=vimeo_logo&owner=83862281.}

According to Egan, Co-Dx has already developed functioning YouTest PCR systems (and begun evaluation deployments of them in various locales around the globe) that deliver multiplexed PCR testing, analysis, and detection.
Specifically, the YouTest PCR system is currently being used to simultaneously test, analyze, and detect for the presence of
- Influenza A (~44,000 strains)
- Influenza B (~12,000 strains), and
- Covid-19,
aka, multiplexing.
In the future the company expects to be able to provide multiplexed analyses and detection of sexually transmitted diseases, Hepatitis B and C, malaria, and other pathogens.
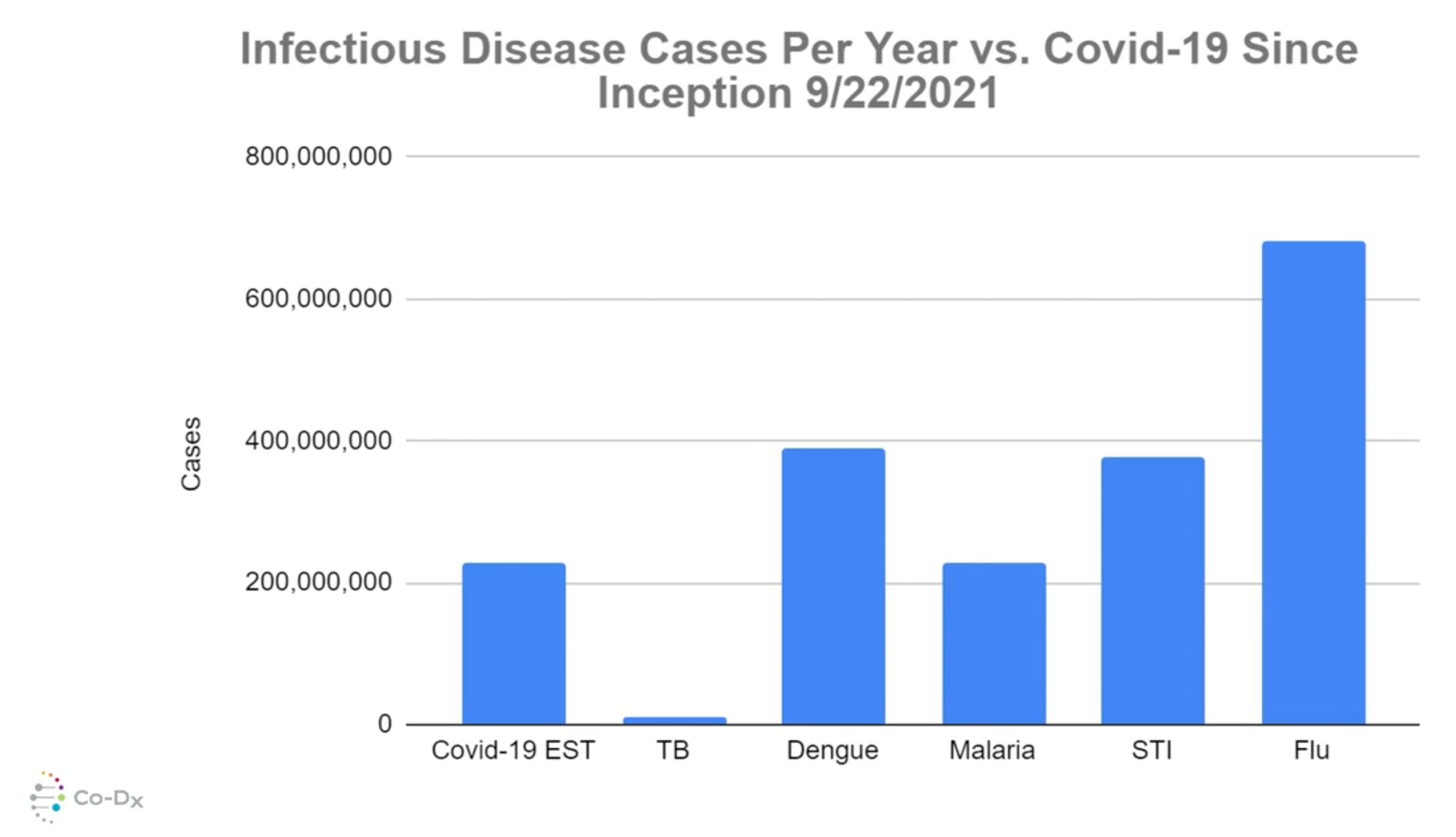
Being able to develop and bring to market less expensive, easy-to-use, multiplexable PCR platforms that deliver fast and accurate results is the real opportunity for Co-Diagnostics, especially when CEO Egan says that there are over 600 million cases of the Common Cold in the United States each year.
That's problematic, he explained, because many of the symptoms of Covid-19 are identical to those of the Common Cold.
But with its approach to PCR multiplexing, Co-Dx expects to be able to simultaneously test and detect up to six to eight disease states with its future products, including the YouTest PCR system.

And that's where Advanced Conceptions and Idaho Molecular come into play, because their combined focus, with Co-Dx, has been in the design and development of the YouTest PCR system and related technologies.
The Multiplying Potential of Global Expertise on Co-Dx
When the acquisitions are completed later this week (assuming all goes according to plan), Dr. Kirk Ririe will become the President of the new, wholly owned Co-Diagnostics subsidiary containing the assets and intellectual property of both Idaho Molecular and Advanced Conceptions.
Additionally, Dr. Carl Wittwer, M.D. will become the Chairman of the Co-Diagnostics Advisory Board.
As it turns out, both Dr. Ririe and Dr. Wittwer have 30-plus years of experience advancing the science of molecular detection, starting with the formation of BioFire Diagnostics and its predecessor companies, BioFire and Idaho Technology (dating back over 30 years ago).
Together, these two professionals have arguably done as much, if not more, than any other two scientists on the planet to advance the understanding and application PCR science.
And now, both will become direct / indirect contributors to future PCR advancements via Co-Diagnostics.
That, to me, is very, very intriguing, both for Co-Dx, but also for the global healthcare community, as well as concerned consumers.
And, now that I think of it, potentially for investors too.